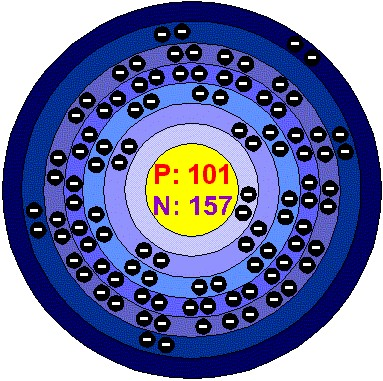
Mendelevium One Hundred First Periodc Table Element
The mendelevium (named after the Russian chemist Dmitri Mendeleev) is a chemical element of symbol Md, atomic number 101 (101 protons and 101 electrons) and atomic mass equal to 258 u. It is metallic, radioactive, transuranic, of the actinide group.
The mendelevium was first synthesized in 1955 by a team of American scientists led by Albert Ghiorso.
History:
The mendelevium was first synthesized by a team of American scientists formed by Albert Ghiorso (team leader), Glenn T. Seaborg, Bernard Harvey, and Greg Choppin in 1955. The team produced the 256Md (half-life of 76 minutes) bombarding einsteinium-253 with alpha particles (helium nuclei) in the "Berkeley Radiation Laboratory" in a cyclotron (60 inches). Element 101 was the ninth transuranic element synthesized.
Main features:
Researchers have demonstrated that mendelevium has a moderate stability when it presents a state of dipositive oxidation (II) besides the tripositive (III) oxidation state characteristic of the actinide elements. Md-256 was used to discover some chemical properties of this element, even in aqueous solution. There is no use of the mendelevium known, even as trace amounts have been obtained so far.
Isotopes:
Mendelevium radioisotopes were identified, the most stable being 258Md with a half-life of 51.5 days, 260Md with a half-life of 31.8 days, and 257Md with a half-life of 5.52 hours. All other radioactive isotopes have half-lives less than 97 minutes, and most of these less than 5 minutes. This element also has 1 meta state, 258mMd (t½ 57 minutes). The atomic masses of mendelevium isotopes range from 245,091 u (245 Mo) to 260,104 u (260 Mo).
The mendelevium was first synthesized in 1955 by a team of American scientists led by Albert Ghiorso.
History:
The mendelevium was first synthesized by a team of American scientists formed by Albert Ghiorso (team leader), Glenn T. Seaborg, Bernard Harvey, and Greg Choppin in 1955. The team produced the 256Md (half-life of 76 minutes) bombarding einsteinium-253 with alpha particles (helium nuclei) in the "Berkeley Radiation Laboratory" in a cyclotron (60 inches). Element 101 was the ninth transuranic element synthesized.
Main features:
Researchers have demonstrated that mendelevium has a moderate stability when it presents a state of dipositive oxidation (II) besides the tripositive (III) oxidation state characteristic of the actinide elements. Md-256 was used to discover some chemical properties of this element, even in aqueous solution. There is no use of the mendelevium known, even as trace amounts have been obtained so far.
Isotopes:
Mendelevium radioisotopes were identified, the most stable being 258Md with a half-life of 51.5 days, 260Md with a half-life of 31.8 days, and 257Md with a half-life of 5.52 hours. All other radioactive isotopes have half-lives less than 97 minutes, and most of these less than 5 minutes. This element also has 1 meta state, 258mMd (t½ 57 minutes). The atomic masses of mendelevium isotopes range from 245,091 u (245 Mo) to 260,104 u (260 Mo).