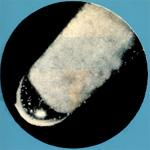
Curium Ninety-sixth Element of the Periodic Table
Curium (named after the couple Pierre and Marie Curie) is a chemical element of the Atomic 96 symbol Cm (96 protons and 96 electrons) with atomic mass 247 u. It is a metallic, synthetic, transuranic element of the actinide group.
It was discovered in 1944 by an American team consisting of Glenn Seaborg, Ralph James, and Albert Ghiorso, produced by bombarding plutonium with alpha particles. Curium-242 and Curium-244 can be used as portable power sources in pacemakers and remote locating instruments.
History:
Curium was first synthesized at the University of California (Berkeley) by Glenn T. Seaborg, Ralph A. James, and Albert Ghiorso in 1944.2 The team named the new curium element named after Marie Curie and her husband Pierre, famous for the discovery of the radio element and its research in radioactivity: As we would like to propose "curium", with symbol Cm. The evidence indicates that element 96 contains seven 5f electrons and is thus analogous to the gadolinium element with its seven 4f electrons in the regular rare earth series. On this basis element 96 is named after the Curies in a manner analogous to the naming of gadolinium, in which the chemist Gadolin was honored.
The element was chemically identified at the Metallurgy Laboratory (now known as the "Argonne National Laboratory") at the University of Chicago. It was the third transuranic element discovered, even being the second in the series. Curium-242 (163 days half-life) and a free neutron were obtained by bombarding a plutonium-239 target with alpha particles on a Berkeley cyclotron.
Louis Werner and Isadore Perlman created a sample of curium-242 at the University of California in 1947 in the form of hydroxide, bombarding americium-241 with neutrons.3 Curium was first produced in elemental form in 1951 by Crane, Wallmann, and Cunningham.4 5
Main Features: Curium is somewhat similar to gadolinium rare earth, but with a more complex crystalline structure. Chemically reactive, it is a silver-white metal (most of its trivalent compounds are slightly yellow), malleable and, due to its high radioactivity, glows in the dark. The element is more electropositive than aluminum.
Curium-248 isotope has been synthesized only in milligram quantities, but curium-242 and curium-244 are produced in multigram quantities, which allows the determination of some of the properties of the element. Curium is obtained in this amount by subjecting plutonium to bombardment of alpha particles. Very small amounts of curium may exist in uranium ore as a product of natural deterioration, but it has never been detected.
Applications:
There are few commercial applications for curium. May be useful in thermoelectric generators. Curium-242 and Cu-244 are used as portable power sources as they can generate around 2 watts of thermal energy per gram. It is used in artificial coronary pacemakers, instruments operating at remote locations on Earth, and in space missions.
Compounds:
Several curium compounds were produced. These include: curium dioxide (CmO2), curium trioxide (Cm2O3), curium bromide (CmBr3), curium chloride (CmCl3), curium tetrafluoride (CmF4) and curium iodide (CmI3).
It was discovered in 1944 by an American team consisting of Glenn Seaborg, Ralph James, and Albert Ghiorso, produced by bombarding plutonium with alpha particles. Curium-242 and Curium-244 can be used as portable power sources in pacemakers and remote locating instruments.
History:
Curium was first synthesized at the University of California (Berkeley) by Glenn T. Seaborg, Ralph A. James, and Albert Ghiorso in 1944.2 The team named the new curium element named after Marie Curie and her husband Pierre, famous for the discovery of the radio element and its research in radioactivity: As we would like to propose "curium", with symbol Cm. The evidence indicates that element 96 contains seven 5f electrons and is thus analogous to the gadolinium element with its seven 4f electrons in the regular rare earth series. On this basis element 96 is named after the Curies in a manner analogous to the naming of gadolinium, in which the chemist Gadolin was honored.
The element was chemically identified at the Metallurgy Laboratory (now known as the "Argonne National Laboratory") at the University of Chicago. It was the third transuranic element discovered, even being the second in the series. Curium-242 (163 days half-life) and a free neutron were obtained by bombarding a plutonium-239 target with alpha particles on a Berkeley cyclotron.
Louis Werner and Isadore Perlman created a sample of curium-242 at the University of California in 1947 in the form of hydroxide, bombarding americium-241 with neutrons.3 Curium was first produced in elemental form in 1951 by Crane, Wallmann, and Cunningham.4 5
Main Features: Curium is somewhat similar to gadolinium rare earth, but with a more complex crystalline structure. Chemically reactive, it is a silver-white metal (most of its trivalent compounds are slightly yellow), malleable and, due to its high radioactivity, glows in the dark. The element is more electropositive than aluminum.
Curium-248 isotope has been synthesized only in milligram quantities, but curium-242 and curium-244 are produced in multigram quantities, which allows the determination of some of the properties of the element. Curium is obtained in this amount by subjecting plutonium to bombardment of alpha particles. Very small amounts of curium may exist in uranium ore as a product of natural deterioration, but it has never been detected.
Applications:
There are few commercial applications for curium. May be useful in thermoelectric generators. Curium-242 and Cu-244 are used as portable power sources as they can generate around 2 watts of thermal energy per gram. It is used in artificial coronary pacemakers, instruments operating at remote locations on Earth, and in space missions.
Compounds:
Several curium compounds were produced. These include: curium dioxide (CmO2), curium trioxide (Cm2O3), curium bromide (CmBr3), curium chloride (CmCl3), curium tetrafluoride (CmF4) and curium iodide (CmI3).