Carbon Sixth element of the Periodic Table
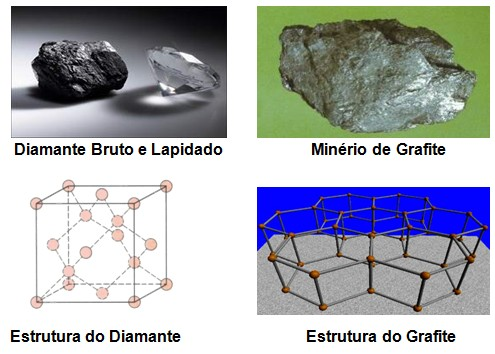
Carbon is undoubtedly an extremely important chemical element because it is indispensable for the existence of life - be it animal and plant - not to mention the mineral compounds constituted by the element in question. The non-metallic tetravalent carbon element, located in family 4A of the periodic table, has atomic number 6 and atomic mass 12, and symbol C.It was discovered in antiquity, its union with other elements for the formation of compounds and molecules is called covalent bond, that is, carbon shares its electrons with the other elements connecting tetrahedrically with which it has electronic affinity, except on some occasions. . Its presence in nature occurs in two allotropic forms: diamond and graphite. Diamond is a covalent solid that has very high hardness and boiling point and the hardest material while graphite is less resistant.
Normally most of the carbon present in nature is in the form of compounds, especially in organic compounds that have the backbone of their chains composed of this element. Carbon is so essential to life that DNA, proteins, and other life-important compounds are formed by carbon chains, among other compounds widely studied by biochemistry.
Organic Chemistry is the branch of chemistry that deals exclusively with the study of carbon and its compounds, this does not mean that there are no inorganic carbon compounds, as we will see below. It is present throughout the animal and plant kingdom forming the essential compounds for life. In minerals, it is in the form of carbonates, carbides and bicarbonates.
The presence of carbon is daily, and it is observed by the compounds whose chemical formulas are formed by a carbonic skeleton such as the cellulose of our clothes and paper, plastics and our foods and even the numbers and weights. The atomic masses refer to the atomic mass of carbon according to the 1977 convention of the IUPAC Atomic Weights Commission. In addition to the diamond and graphite mentioned above, carbon presents another allotropic form discovered in 1985 called fullerene with molecules forming structures with 60 C atoms, named after the architect R. Buckminister Fuller.
Important Information: Symbol: C Atomic Mass: 12 u Atomic Number: 6 Melting Point: 3550 ° C Boiling Point: 4289 ° C Allotropic forms: Diamond and graphite. Electronic Configuration: 1s², 2s², 2p² Hybridization: sp³
Some Carbon Compounds: CH4, Na2CO3, C2H6, C2H5OH, CaCl2
Bibliography: http://nautilus.fis.uc.pt/ http://www.cdcc.sc.usp.br/ CHEMISTRY - A UNIVERSITY COURSE
/ Mahan, Bruce M. Myers, Rollie J.- 2002 - 4th ed. James Brady, Humiston Gerard E. GENERAL CHEMISTRY - VOL. 2 - 2nd EDITION