The Periodic Table of Chemical Elements
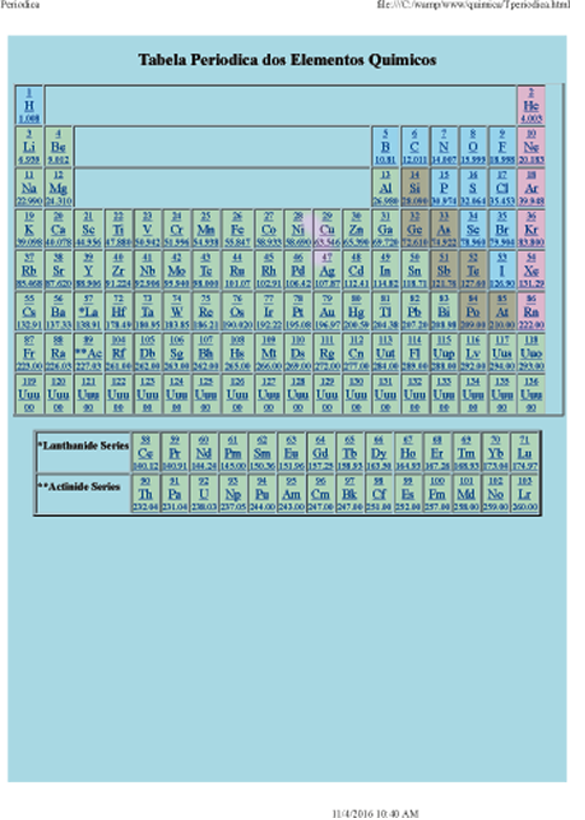
The Periodic Table
The periodic table is a systematic arrangement of the chemical elements ordered by their atomic numbers, electronic configuration, and recurrence of the periodic properties.
This ordering shows periodic trends, such as elements with similar behaviors in the same column. It also shows four rectangular blocks with similar chemical properties.
In general, inside a line (period) the elements are metallic in the left and nonmetallic in the right.
The rows in the table are called periods; the columns are called groups.
Elements with the atomic number of 95 to 136 were only synthesized in laboratory or nuclear reactors. The synthesis of elements with larger atomic numbers has been sought.
Several synthetic or naturally occurring radioactive elements have also been produced in laboratories
.

John Newlands
History:
The history of the periodic table reflects over a century of growth in the understanding of chemical properties. The most important event in its history occurred in 1869, when the table was published by Dmitri Mendeleev, who built upon earlier discoveries by scientists such as Antoine-Laurent de Lavoisier and John Newlands, but who is nevertheless generally given sole credit for its development.

Dmitri Mendeleev
Some important definitions
Atomic Number :
The atomic number or proton number of a chemical element is the number of protons found in the nucleus of an atom. It is identical to the charge number of the nucleus. The atomic number uniquely identifies a chemical element. In an uncharged atom, the atomic number is also equal to the number of electrons.
Mass Number:
Mass number, in nuclear physics, or chemisty the sum of the numbers of protons and neutrons present in the nucleus of an atom. The mass number is commonly cited in distinguishing among the isotopes of an element, all of which have the same atomic number (number of protons) and are represented by the same literal symbol; for example, the two best known isotopes of uranium (those with mass numbers 235 and 238) are designated uranium-235 (symbolized 235U) and uranium-238 (238U).
Isotopes Elements:
Each of two or more forms of the same element that contain equal numbers of protons but different numbers of neutrons in their nuclei, and hence differ in relative atomic mass but not in chemical properties; in particular, a radioactive form of an element.



